Bone Loss
Estrogen deficiency has been well established as a cause of bone loss. This loss can be noted for the first time when menstrual cycles become irregular in the perimenopause. From 1.5 years before the menopause to 1.5 years after menopause, spine bone mineral density (BMD) has been shown to decrease by 2.5% per year, compared with a premenopausal loss rate of 0.13% per year. Loss of trabecula bone (spine) is greater with estrogen deficiency than is loss of cortical bone.
Postmenopausal bone loss leading to osteoporosis is a substantial healthcare problem. In white women, 35% of all postmenopausal women have been estimated to have osteoporosis based on BMD. Further, the lifetime fracture risk for these women is 40%. The morbidity and economic burden of osteoporosis is well documented. Interestingly, there are data to suggest that up to 19% of Caucasian men also have osteoporosis.
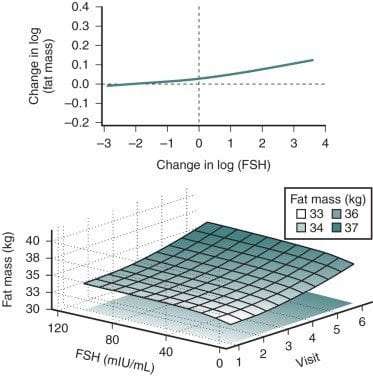
Bone mass is substantially affected by sex steroids through classic mechanisms to be described later in this chapter. Attainment of peak bone mass in the late second decade ( Fig. 14.22 ) is key to ensuring that the subsequent loss of bone mass with aging and estrogen deficiency does not lead to early osteoporosis. Estradiol, together with GH and insulin-like growth factor-1, acts to double bone mass at the time of puberty, beginning the process of attaining peak bone mass. Postpubertal estrogen deficiency (amenorrhea from various causes) substantially jeopardizes peak bone mass. Adequate nutrition and calcium intake are also key determinants. While estrogen is of predominant importance for bone mass in both women and men, testosterone is important in stimulating periosteal apposition; as a result, cortical bone in men is larger and thicker.

ERs are present in osteoblasts, osteoclasts, and osteocystes. Both ERα and ERβ are present in cortical bone, while ERβ predominates in cancellous or trabecular bone. However, the more important actions of estradiol are believed to be mediated via ERα.
Estrogens suppress bone turnover and maintain a certain rate of bone formation. Bone is remodeled in functional units, called bone multicenter units (BMUs), where resorption and formation should be in balance. Multiple sites of bone go through this turnover process over time. Estrogen decreases osteoclasts by increasing apoptosis and thus reduces their lifespan. The effect on the osteoblast is less consistent, but E 2 antagonizes glucocorticoid-induced osteoblast apoptosis. Estrogen deficiency increases the activities of remodeling units, prolongs resorption, and shortens the phase of bone formation ; it also increases osteoclast recruitment in BMUs, thus resorption outstrips formation.
The molecular mechanisms of estrogen action on bone involve the inhibition of production of proinflammatory cytokines including interleukin-1, interleukin-6, and tumor necrosis factor, which inhibits bone resorption. Receptor-activation of nuclear factor kappa-B ligand (RANKL) is responsible for osteoclast differentiation and action. A scheme for how all these factors interact has been proposed by Riggs ( Fig. 14.23 ).

In women, Riggs has suggested that bone loss occurs in two phases. In the first phase, with declining estrogen at the onset of menopause there is an accelerated phase of bone loss; this loss is predominantly of cancellous or trabecula bone. Here 20% to 30% of cancellous bone and 5% to 10% of cortical bone can be lost in a short span of 4 to 8 years. Thereafter, a slower phase of loss (1% to 2% per year) ensues where, proportionately, more cortical bone is lost. This phase is thought to be induced primarily by secondary hyperparathyroidism. The first phase is also accentuated by the decreased influence of stretching or mechanical factors, which generally promote bone homeostasis, as a result of estrogen deficiency.
Genetic influences on bone mass are more important for attainment of peak bone mass (heritable component, 50% to 70%) than for bone loss. Polymorphisms of the vitamin D receptor gene, TGFβ gene, and the Spl-binding site in the collagen type 1 AI gene have all been implicated as being important for bone mass.
While testosterone is important for bone formation and stimulation of bone mass, even in men estrogen action is of paramount importance. Bone mass was shown to increase in an aromatase-deficient man upon estrogen administration.
Bone mass can be detected by a variety of radiographic methods ( Table 14.2 ). Dual energy x-ray absorptiometry scans have become the standard of care for detection of osteopenia and osteoporosis. By convention, the T score is used to reflect the number of standard deviations of bone loss from the peak bone mass of a young adult. Osteopenia is defined by a T score of −1 to −2.5 standard deviations; osteoporosis is defined as greater than 2.5 standard deviations. Since bone mass does not completely reflect bone strength, which is really what matters in terms of fracture risk, several approaches have been made to assess bone strength. An assessment of biochemical bone turnover (discussed later in the chapter), in addition to bone mass, is deemed important. One newer approach is to assess bone microarchitecture, a so-called virtual bone biopsy, by using a high-resolution (HR) peripheral (p) quantitative computed tomography ( Fig. 14.24 ). This technique has been used to assess bone strength and formation in boys and girls to assess sex differences in bone growth and strength, and the risk of fracture. Various biochemical assays are also available to assess bone resorption and formation in both blood and urine ( Table 14.3 ). At present, serum markers appear to be most useful for assessing changes with antiresorptive therapy. Biochemical assays can provide some functional information, which may be helpful in assessing bone strength, and can reflect changes in bone resorption/formation more rapidly than imaging studies. However, these assays do not correlate well with bone density measurements.
| Technique | Anatomical Site of Interest | Precision in vivo (%) | Examination and Analysis Time (min) | Estimated Effective Dose Equivalent (µSv) |
|---|---|---|---|---|
| Conventional radiography | Spine, hip | NA | <5 | 2000 |
| Radiogrammetry | Hand | 1–3 | 5–10 | <1 |
| Radiographic absorptiometry | Hand | 1–2 | 5–10 | <1 |
| Single x-ray absorptiometry | Forearm, heel | 1–2 | 5–10 | <1 |
| Dual x-ray absorptiometry | Spine, hip, forearm, total body | 1–3 | 5–20 | 1–10 |
| Quantitative computed tomography | Spine, forearm, hip | 2–4 | 10–15 | 50–100 |
| Quantitative ultrasound | Heel, hand, lower leg | 1–3 | 5–10 | None |

| Marker | Specimen |
|---|---|
| Bone Resorption Markers | |
| Cross-linked N-telopeptide of type 1 collagen (NTX) | Urine, serum |
| Cross-linked C-telopeptide of type 1 collagen (CTX) | Urine (αα and ββ forms) |
| Serum (ββ form) | |
| MMP-generated telopeptide of type 1 collagen (ICTP or CTX-MMP) | Serum |
| Deoxypyridinoline, free and peptide bound (fDPD, DPD) | Urine, serum |
| Pyridinoline, free and peptide bound (fPYD, PYD) | Urine, serum |
| OHP | Urine |
| GylHyl | Urine, serum |
| HelP | Urine |
| Tartrate-resistant acid phospharase | Serum, plasma |
| 5b Isoform specific for osteoclasts (TRACP 5b) | |
| Cath K | Urine, serum |
| uOC | Urine |
| Bone Formation Markers | |
| OC | Serum |
| Procollagen type 1 C-terminal propeptide (PICP) | Serum |
| Procollagen type 1 N-terminal propeptide (PINP) | Serum |
| Bone-specific alkaline phosphatase (bone ALP) | Serum |
There are now many agents that can prevent osteoporosis. The use of estrogen will depend on whether or not there are other indications for estrogen treatment and whether there are contraindications. Estrogen has been shown to reduce the risk of osteoporosis as well as to reduce osteoporotic fractures. In the WHI study, hip fractures, as well as all fractures, were reduced with conjugated equine estrogens (CEE) and medroxyprogesterone acetate (MPA), and CEE alone, and this occurred in a nonosteoporotic population. Indeed, in a cohort of women who were followed after stopping hormones when the results of the WHI study were published, there was an increased risk of hip fracture and lower bone density compared to those women who continued therapy.
A dose equivalent to 0.625 mg of CEE was once thought to prevent osteoporosis, but we now know that lower doses (0.3 mg of CEE or its equivalent) in combination with progestogens are able to prevent bone loss, although there are no data on fractures. Whether or not the addition of progestogens, by stimulating bone formation, increases bone mass over that of estrogen alone is unclear. The androgenic activity of certain progestogens such as northindrone acetate also has been suggested to play a role.
Selective estrogen receptor modulators (SERMs) , such as raloxifene, droloxifene, and tamoxifen, have all been shown to decrease bone resorption. Raloxifene has been shown to decrease vertebral fractures in a large prospective trial. Tibolone has also been shown to be an effective treatment for osteoporosis. Tibolone (not marketed in the United States) has SERM-like properties, but it is not specifically a SERM because it has mixed estrogenic, antiestrogenic, androgenic, and progestogenic properties due to its metabolites. The drug does not cause uterine or breast cell proliferation and is beneficial for vasomotor symptoms. It prevents osteoporosis and has been shown to be beneficial in the treatment of established osteoporosis.
Bisphosphonates have been shown to have a significant effect on the prevention and treatment of osteoporosis. With this class of agents (etidronate, alendronate, residronate, ibandronate, and zoledronic acid) there is incorporation of the bisphosphonate with hydroxyapetite in bone, which increases bone mass. The skeletal half-life of bisphosphonates in bone can be as long as 10 years. Most data have been derived with alendronate, which, at a dosage of 5 mg daily (35 mg weekly) prevents bone loss; at 10 mg daily (70 mg weekly), alendronate is an effective treatment for osteoporosis, with evidence available that this treatment reduces vertebral and hip fractures. Ibandronate is available as a monthly pill (150 mg) and by injection (3 mg) every 3 months. It has primary efficiency for vertebral fracture protection. Zoledronic acid, 5 mg, is available as an intravenous therapy, with infusion over 15 minutes.
There has been some concern over this class of very powerful bone resorption agents causing fractures of the long bones such as the femur because of “brittle” bone. This only occurs with prolonged use of at least 7 years, and the incidence is in the range of 3.2 to 50 per 100,000 women. Osteonecrosis of the jaw has also been cited as a concern, but this mainly occurs in the presence of poor dental health, and is rare with an incidence in the range of 1/10,000 women. With long-term therapy, atrial fibrillation, as an adverse event also occurs, although this too is rare. These adverse effects appear to be a “class” effect of bisphosphonates. Nevertheless, while all these findings are rare events, there are no good data for prolonged treatment of 10 or more years. For these and other reasons, bisphosphonates are not the drugs of choice in younger women prior to natural menopause and should not be used in women who wish to conceive.
Calcitonin, 50 IU subcutaneous injections daily or 200 IU intranasally, has been shown to inhibit bone resorption. Vertebral fractures have been shown to decrease with calcitonin therapy. However, long-term effects have not been established.
Fluoride has been used for women with osteoporosis because it increases bone density. Currently, a lower dose (50 µg daily) of slow-release sodium fluoride does not seem to cause adverse effects (gastritis) and has efficacy in preventing vertebral fractures.
Denosumab is a monoclonal antibody targeting RANKL, which is secreted by osteoblasts and causes bone resorption (see Fig. 14.22 ). Thus it is an antiresorptive agent with significant potency. Denosumab 60 mg is administered subcutaneously every 6 months, and while it has a preventative role it is usually considered to be a second-line treatment for difficult-to-treat cases. Denosumab has efficacy both at the vertebrae and the hip, but unlike the bisphosphonates, is shorter acting and wears off quickly rather than being bound to the bone with a long half-life in the case of bisphosphonates. Also it is devoid of the other side effects of bisphosphonates (fractures, osteonecrosis, etc.) described above. Because of this profile and its benefit at the hip as well as the spine, denosumab has emerged as a popular therapy. Nevertheless, as a newer form of immune therapy, long-term potential consequences are not known.
An inhibitor of cathepsin K, odanacatib, also has a significant effect on decreasing bone resorption and has been successful in clinical trials at vertebral and nonvertebral bone sites. However, odanacatib is not yet available for clinical use.
Intermittent parathyroid hormone (PTH 1-34, teriparatide) is an agent that increases bone mass in women with osteoporosis. In a randomized trial lasting 3 years, average bone density increased in the hip and spine with fewer fractures observed. This therapy is available in the United States, at a standard dose of 20 µg/day injected subcutaneously for no longer than 18 months. It is expensive and is reserved for women who are difficult to treat and have a history of fractures.
Another agent, which has the ability to increase bone formation, is a monoclonal antibody against sclerosin. Sclerosin is an inhibitor of normal bone formation, which activated the Wnt signaling system. While this monoclonal antibody, romosozumab, has shown great efficacy in increasing bone formation, its cardiovascular risk profile has not been acceptable; and it is unlikely that this drug will be available for clinical use.
Adjunctive measures for the prevention of osteoporosis are calcium, vitamin D, and exercise. Calcium with vitamin D treatment has been shown to increase bone only in older individuals. These modalities alone are not thought to be effective for the treatment of osteoporosis. A woman’s total intake of elemental calcium should be 1200 to 1500 mg daily if no agents are being used to inhibit resorption, 400 to 800 IU of vitamin D should also be ingested. Levels of serum 25 hydroxy vitamin D have been found to be abnormally low (<20 ng/mL) in a large population of women, particularly in geographic regions of less sunlight exposure. Exercise has been shown to be beneficial for building muscle and bone mass and for reducing falls. Guidelines regarding the management of osteoporosis were published by the National Osteoporosis Foundation, last updated in 2013 ( www.nof.org ). An update analysis has suggested that 37% of women are candidates for treatment to prevent fractures. Also, the World Health Organization (WHO) has produced guidelines for assessing an individual’s risk of osteoporosis based on history, anthropometry, and BMD. This new paradigm, called the Fracture Risk Assessment Tool, may be obtained at www.shef.ac.uk/FRAX .
This content was originally published here.